
October 23, 2017
LA JOLLA—Salk Institute scientists will lead a multimillion-dollar, five-year initiative to revolutionize our understanding of the human brain by systematically identifying and cataloging cell types across the mammalian brain, the National Institutes of Health has announced. The effort, which is part of the Brain Research through Advancing Innovative Neurotechnologies (BRAIN) Initiative®, will be co-led by Salk Professors Joseph Ecker and Ed Callaway. Researchers from USC and UC San Diego will also participate in the collaboration.
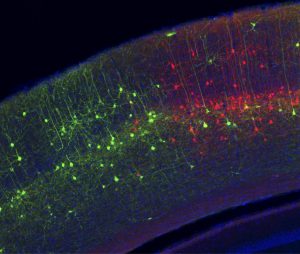
Click here for a high-resolution image.
Credit: Marina Garrett
“Developing an atlas of brain cell types is an incredibly ambitious undertaking but one that is critical to uncover brain function,” says Salk President Elizabeth Blackburn. “With this generous grant, we will be able to better understand the organ that not only makes us who we are, but whose dysfunction—from mental illness to the degeneration of Alzheimer’s disease—causes some of the most acute kinds of human suffering.”
The brain is not just made up of one type of neuron, but many types of cells that scientists are just beginning to identify. Pinpointing these different cells can help reveal how the brain works—and what happens when it doesn’t—but scientists lack a systematic catalog that identifies cell types across the entire brain. They also lack appropriate means for genetically targeting the majority of cell types for research into therapeutic uses, particularly in humans.
To address these limitations, Ecker and Callaway, along with Salk colleagues and other collaborators, propose a series of integrated experiments to identify, catalog and characterize brain cell types in mice via molecular signatures and then link those cells types to their inputs and outputs. Of particular use will be a method pioneered by the Callaway lab to use modified rabies virus to trace connections between nerve cells. The project, called the Center for Epigenomics of the Mouse Brain Atlas (CEMBA), will require collecting data from over 100 brain regions covering the entire mouse brain and associating those cell types with anatomical features that may be common to nervous systems generally and therefore subject to similar methods of monitoring and manipulation in other species.
“One of the first steps in understanding how any complex system works, from a car to a computer, is to come up with a parts list,” says Joseph Ecker, Howard Hughes Medical Institute Investigator and director of Salk’s Genomic Analysis Laboratory. “In the case of the brain, we have only just begun to identify the myriad cell types we know must exist. Once we have a more complete understanding of what the parts are, we will be able to investigate their function and, perhaps more importantly, their dysfunction. This grant represents a big leap forward in achieving those goals.”

Click here for a high-resolution image.
Credit: Salk Institute
The Salk center is focused on single-cell epigenetics—the chemical changes to DNA that are responsible for controlling the differences in gene expression that make each brain cell type unique. The team expects that this information will be particularly useful for creating tools to target these same cell types in future studies of brain circuit function.
“We are excited to be part of a collaborative team composed of several different NIH-funded Centers that will make up the BRAIN Initiative Cell Census Network (BICCN),” says Salk Professor Ed Callaway, who holds the Audrey Geisel Chair in Biomedical Science. “Recent advances in single-cell genomic technology have made it possible to genetically interrogate millions of individual brain cells. This work will lay a foundation for the study of brain circuits with impact comparable to what sequencing the genome had on cell and molecular biology.”
The collaboration includes:
For more information, please see the funding announcement at https://grants.nih.gov/grants/guide/rfa-files/RFA-MH-17-225.html. This project is supported by NIMH/NIH Cooperative Agreement Number U19MH11483.
Office of Communications
Tel: (858) 453-4100
press@salk.edu
The Salk Institute is an independent, nonprofit research institute founded in 1960 by Jonas Salk, developer of the first safe and effective polio vaccine. The Institute’s mission is to drive foundational, collaborative, risk-taking research that addresses society’s most pressing challenges, including cancer, Alzheimer's disease, and agricultural vulnerability. This foundational science underpins all translational efforts, generating insights that enable new medicines and innovations worldwide.