Associate Professor
Clayton Foundation Laboratories for Peptide Biology
Pioneer Fund Developmental Chair

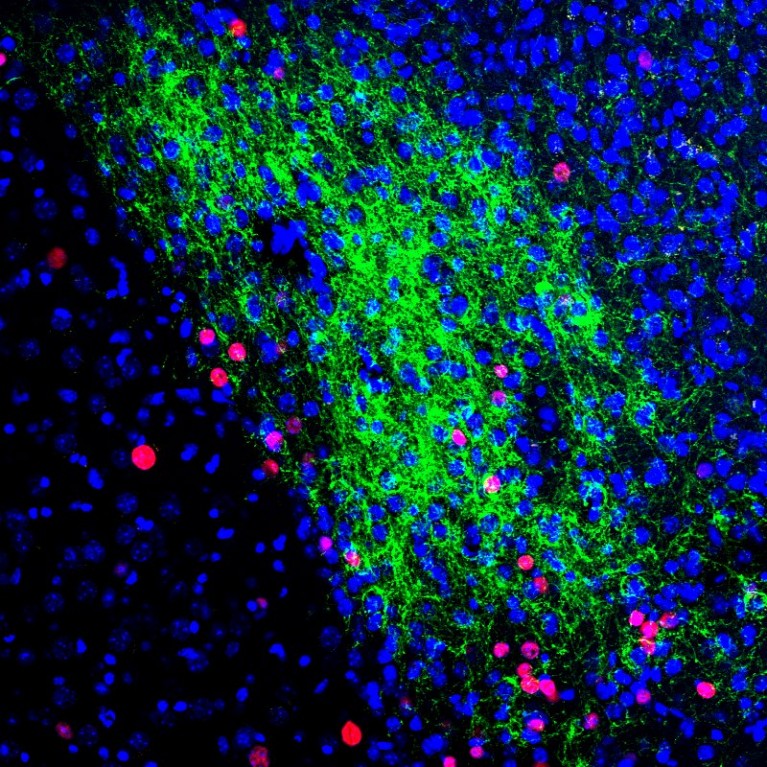
To escape from potentially harmful situations, the brain has a built-in alarm system. This system first recognizes environmental threats using its sensory systems, and then issues orders to change physiology, metabolism, behaviors and emotions to avoid these threats. However, just as with house or car security technology, the biological system can malfunction and generate false alarms. This hypersensitivity to otherwise normal sensory stimuli is linked to neuropsychiatric disorders, such as PTSD, panic disorders, anxiety disorder, chronic pain disorder, schizophrenia and autism spectrum disorders. Despite its critical involvement for survival and for disease pathogenesis, much remains unknown about how the brain’s alarm system works, mainly due to the lack of appropriate tools that show specific neural circuits responsible for behaviors.
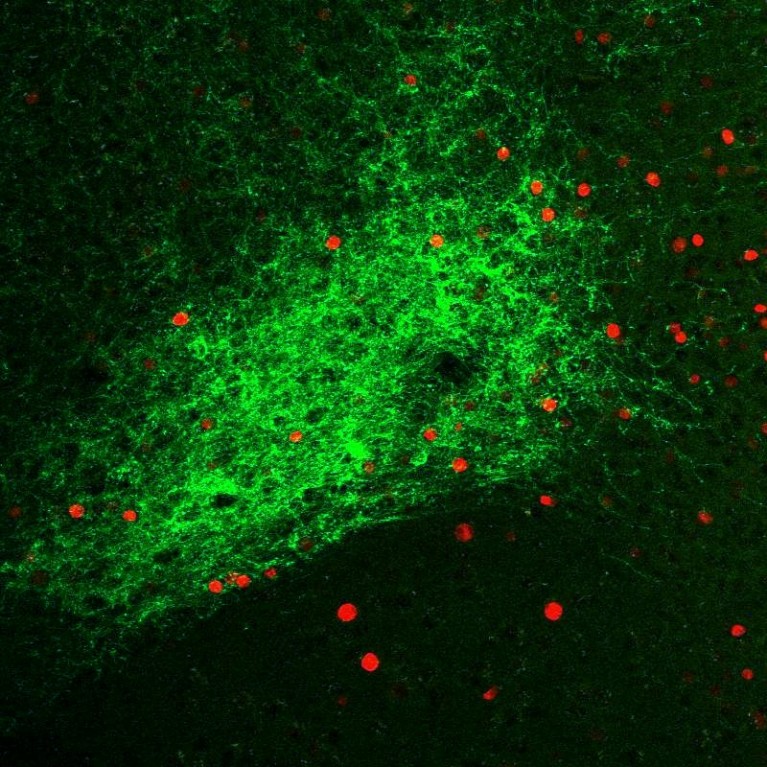
Recent discoveries show that each type of neuron has its own chemical markers that determine its functional role in the brain. Sung Han examines specific markers called neuropeptides that influence the brain’s alarm system. The dysfunction of neuropeptide signaling has been directly linked to a variety of human disorders, ranging from neuropsychiatric disorders to metabolic syndromes. Although hundreds of neuropeptides are identified, their functional roles in the brain are not fully understood. To better understand how neuropeptide signaling encodes and processes sensory threats, Han is unraveling individual neural circuits using the latest techniques in the field of neuroscience, including optogenetics, chemogenetics, electrophysiology and in vivo and in vitro functional imaging techniques, among others. Understanding the neural circuitry of the brain’s alarm system will provide insights for developing therapeutic interventions for a host of neuropsychiatric disorders with sensory abnormalities.
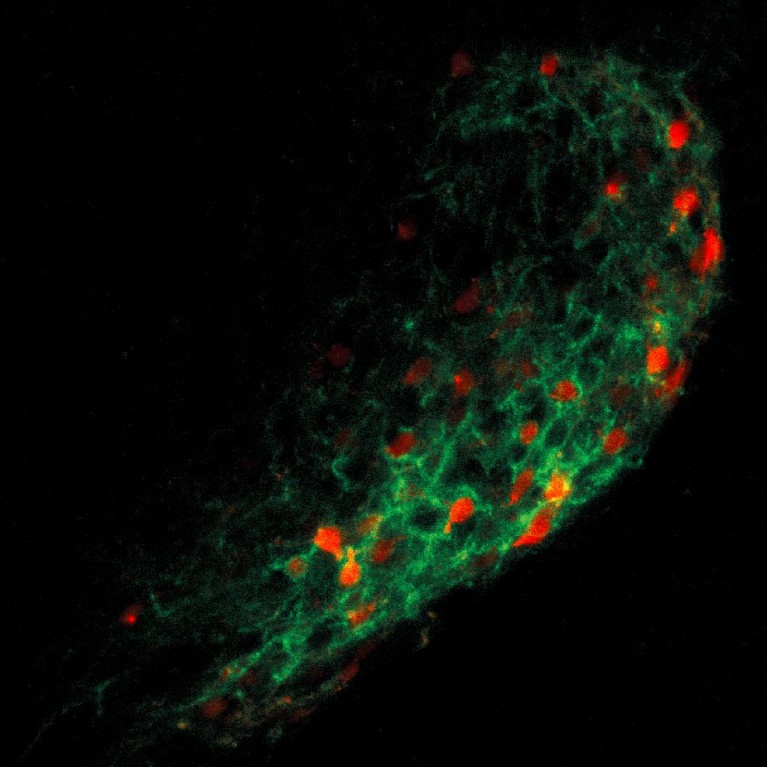
Han discovered that the mutation of a gene that can make neurons excitable leads to autism spectrum disorder by decreasing the balance between excitatory and inhibitory signals in the brain.
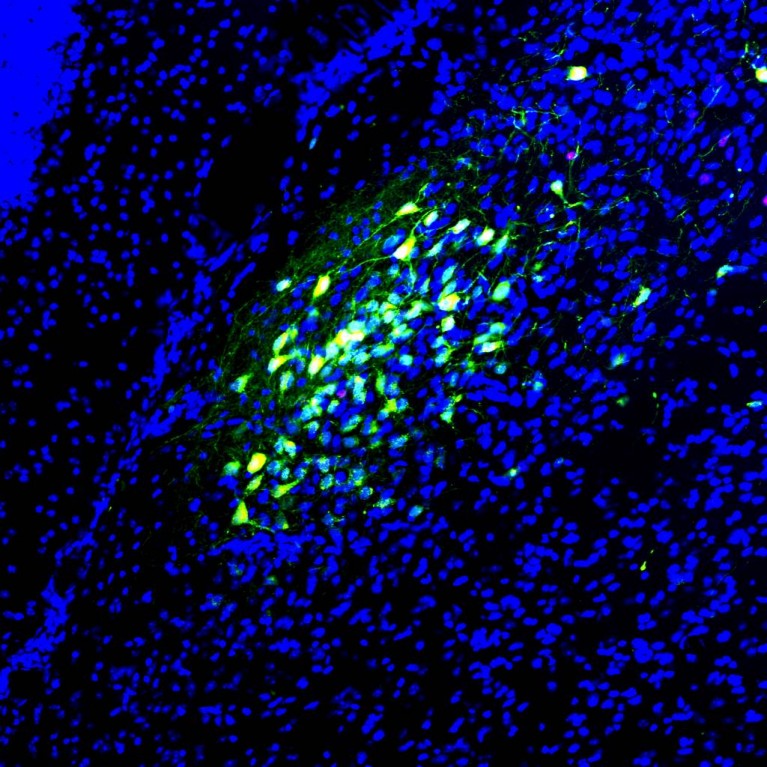
He also found that a very low dose of benzodiazepine, a tranquilizer drug, relieves symptoms of autism spectrum disorders, including social impairment, repetitive behaviors and cognitive deficits, in multiple mouse models by rebalancing the ratio of neurons activated and inhibited in the brain.
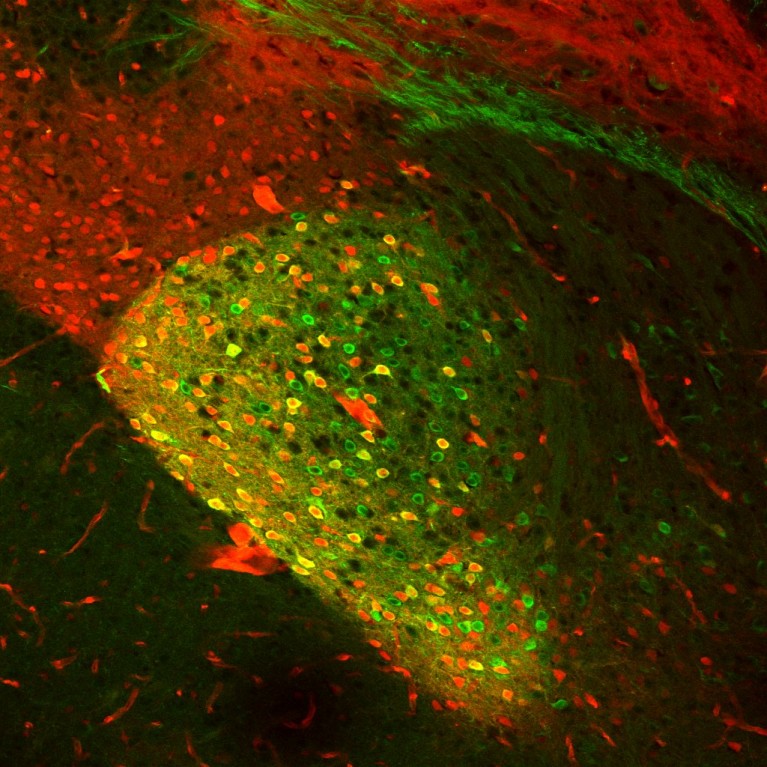
Han found a neural circuit that contains neurons expressing the neuropeptide calcitonin gene-related peptide (discovered by Salk’s Ronald Evans), which mediates emotional and motivational aspects of pain from the periphery to the brain.
BS, Genetic Engineering, Kyungpook National University, Korea
MS, Neuroscience, Pohang University of Science and Technology, Korea
PhD, Neurobiology & Behavior, University of Washington, Seattle