Professor
Waitt Advanced Biophotonics Center
Françoise Gilot-Salk Chair

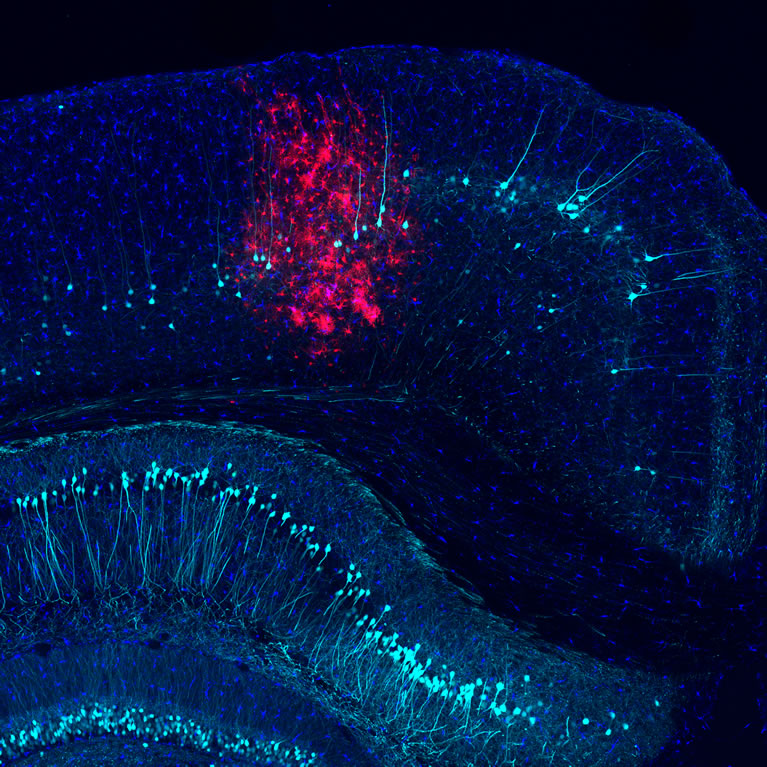
The human central nervous system (CNS), which includes the brain and spinal cord, consists of an incredibly diverse set of cells, and each cell type carries out highly specialized functions in cellular networks of dazzling complexity. While much research has focused on understanding the circuits formed by neurons, brain cells called glia are equally pervasive and account for roughly an equal number of cells in the human CNS. Glial cells were long believed to play merely passive, supportive roles in CNS function. However, it is now clear that glial cells make crucial contributions to CNS formation, operation and adaptation. Additionally, glial cells are involved in practically all CNS injuries and diseases, including viral and bacterial infection, Alzheimer’s and Parkinson’s disease, spinal cord injury, cancer and stroke. This makes glia promising targets for novel therapeutic interventions.
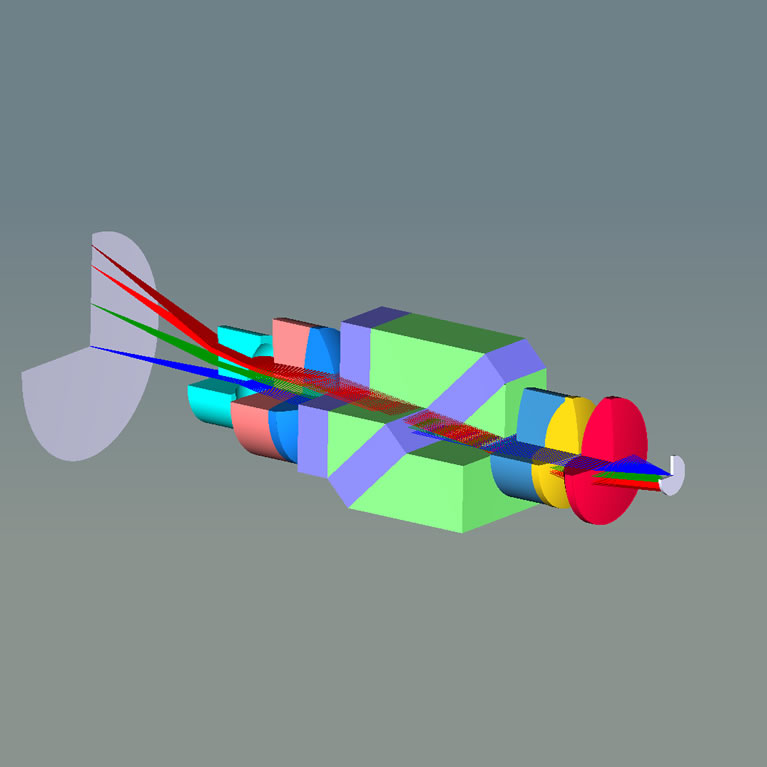
Axel Nimmerjahn has spearheaded the development of new microscopy techniques to visualize the structural and functional dynamics of glial cells and their bi-directional interaction with other cells. To enable cellular-resolution measurements under naturalistic conditions, his lab has worked to shrink the size of microscopes to make them wearable. Their tiny microscopes weigh less than 2.5 grams, are only a few millimeters in size, and have allowed the team to reveal how cellular activity encodes sensory and motor information. Additionally, they have created new tools for cell type-specific staining and genetic manipulation and for analysis of large-scale imaging data. This has allowed them to address long-standing questions regarding the role of glial cells in the intact healthy or diseased CNS (see below). Resolving these fundamental questions has broad implications for our understanding of CNS function and the treatment of neuroinflammatory and neurological disorders.
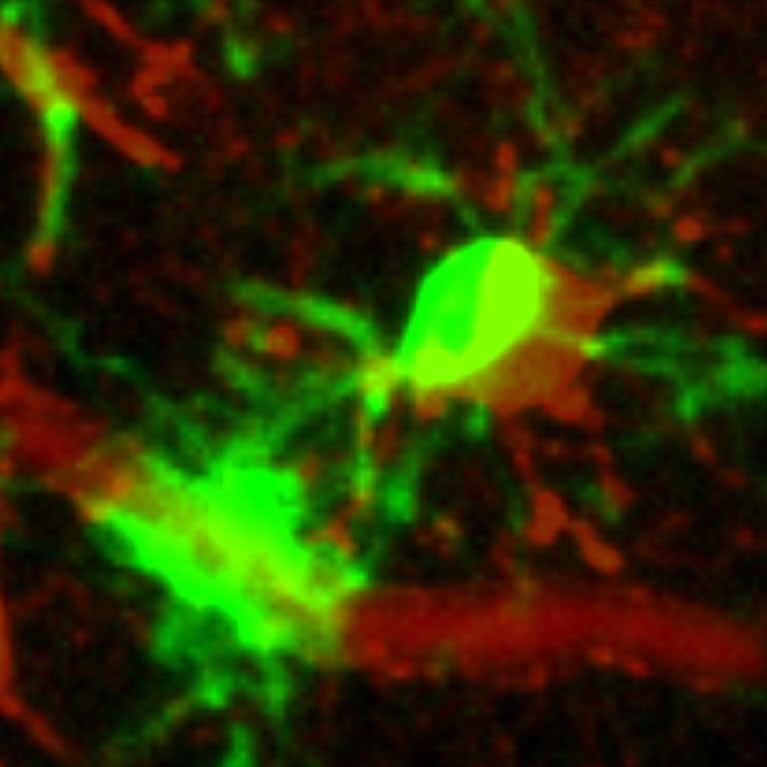
Nimmerjahn discovered that microglia, the resident immune cells in the CNS, continuously survey the cellular environment with their fine branches. He showed that through this behavior, microglia provide the first line of defense against tissue injury and infection, and he identified mechanisms that regulate this inflammatory response (patent pending).
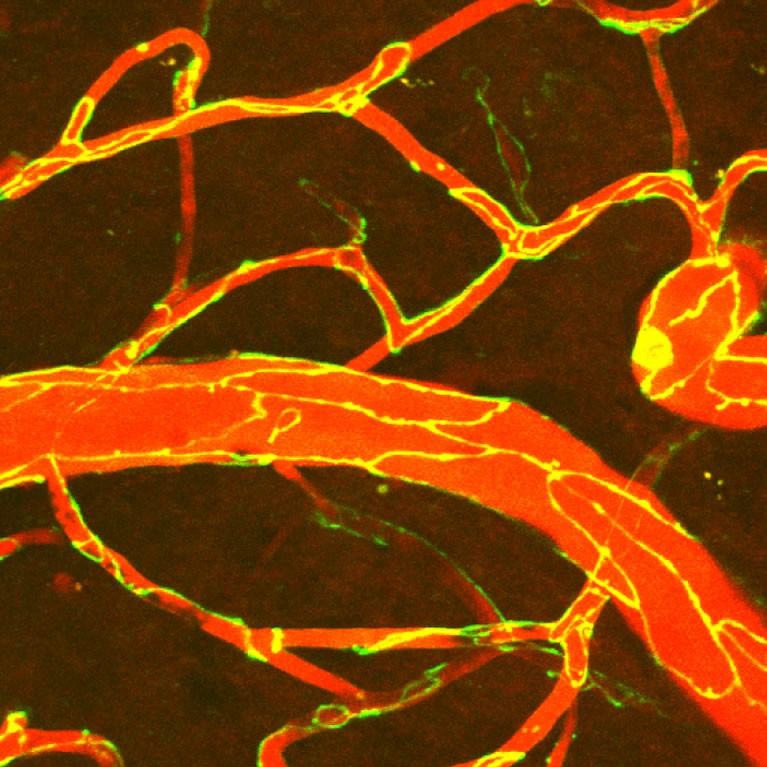
Nimmerjahn’s lab used cutting-edge microscopy approaches to visualize the blood-brain barrier (BBB) breakdown after stroke. His team found that stepwise impairment of different cellular mechanisms accounts for the BBB deficits in stroke. The findings could lead to new ways to treat the disease.
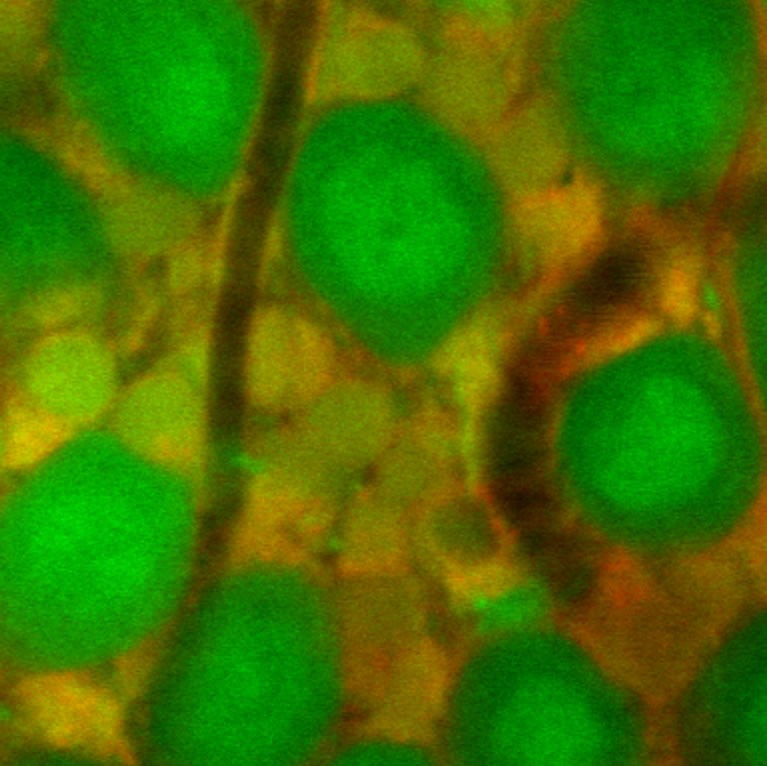
Nimmerjahn uncovered that astroglia, a major regulatory cell type in the CNS, respond to painful stimuli with large-scale coordinated excitation suited to initiate macroscopic changes in CNS network dynamics, and showed how antinociceptive drugs disrupt this activity. This makes astroglia potential new targets for treatment of painful conditions.
MS, Physics, Max Planck Institute for Medical Research/University of Heidelberg, Germany
PhD, Physics, Max Planck Institute for Medical Research/University of Heidelberg, Germany
Postdoctoral Fellow, Biology and Applied Physics, Stanford University