

May 11, 2015
Salk scientists find a single molecule that controls the fate of mature sensory neurons
Salk scientists find a single molecule that controls the fate of mature sensory neurons
LA JOLLA–Scientists at the Salk Institute have discovered that the role of neurons–which are responsible for specific tasks in the brain–is much more flexible than previously believed.
By studying sensory neurons in mice, the Salk team found that malfunction of a single molecule can prompt the neuron to make an “early-career” switch, changing a neuron originally destined to process sound or touch, for example, to instead process vision.
The finding, reported May 11, 2015 in PNAS, will help neuroscientists better understand how brain architecture is molecularly encoded and how it can become miswired. It may also point to ways to prevent or treat human disorders (such as autism) that feature substantial brain structure abnormalities.
“We found an unexpected mechanism that provides surprising brain plasticity in maturing sensory neurons,” says the study’s first author, Andreas Zembrzycki, a senior research associate at the Salk Institute.
The mechanism, a transcription factor called Lhx2 that was inactivated in neurons, can be used to switch genes on or off to change the function of a sensory neuron in mice. It has been known that Lhx2 is present in many cell types other than in the brain and is needed by a developing fetus to build body parts. Without Lhx2, animals typically die in utero. However, it was not well known that Lhx2 also affects cells after birth.
“This process happens while the neuron matures and no longer divides. We did not understand before this study that relatively mature neurons could be reprogrammed in this way,” says senior author Dennis O’Leary, Salk professor and holder of the Vincent J. Coates Chair in Molecular Neurobiology. “This finding opens up a new understanding about how brain architecture is established and a potential therapeutic approach to altering that blueprint.”

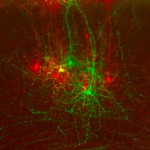
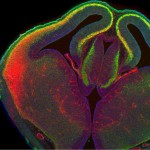

Scientists had believed that programming neurons was a one-step process. They thought that the stem cells that generate the neurons also programmed their functions once they matured. While this is true, the Salk team found that another step is needed: the Lhx2 transcription factor in mature neurons then ultimately controls the fate of the neuron.
In the mouse study, the scientists manipulated Lhx2 to make the switch in neuronal fate shortly after birth (when the mouse neurons are fully formed and considered mature). The team observed that controlling Lhx2 let them instruct neurons situated in one sensory area to process a different sense, thus enlarging one region at the expense of the other. The scientists don’t know yet if targeting Lhx2 would allow neurons to change their function throughout an organism’s life.
“This study provides proof that the brain is very plastic and that it responds to both genetic and epigenetic influences well after birth,” says O’Leary. “Clinical applications for brain disorders are a long way away, but we now have a new way to think about them.”
“Since this study was conducted in mice, we don’t know the time frame in which Lhx2 would be operating in humans, but we know that post-birth, neurons in a baby’s brain still have not settled into their final position–they are still being wired up. That could take years,” Zembrzycki says.
However, the findings may be an ingredient that contributes to the success of early intervention in some very young children diagnosed with autism, adds Zembrzycki. “The brain’s wiring is determined genetically as well as influenced epigenetically by environmental influences and early intervention preventing brain miswiring may be an example of converging genetic and epigenetic mechanisms that are controlled by Lhx2.”
Authors of the work are Andreas Zembrzycki, Carlos G. Perez-Garcia, and Dennis D. M. O’Leary, all of the Salk Institute for Biological Studies; and Chia-Fang Wang and Shen-Ju Chou, of the Institute of Cellular and Organismic Biology, Academia Sinica, in Taiwan.
The work was funded by the National Institutes of Health and a grant from the National Science Council, Taiwan.
About the Salk Institute for Biological Studies:
The Salk Institute for Biological Studies is one of the world’s preeminent basic research institutions, where internationally renowned faculty probes fundamental life science questions in a unique, collaborative and creative environment. Focused both on discovery and on mentoring future generations of researchers, Salk scientists make groundbreaking contributions to our understanding of cancer, aging, Alzheimer’s, diabetes and infectious diseases by studying neuroscience, genetics, cell and plant biology and related disciplines.
Faculty achievements have been recognized with numerous honors, including Nobel Prizes and memberships in the National Academy of Sciences. Founded in 1960 by polio vaccine pioneer Jonas Salk, MD, the Institute is an independent nonprofit organization and architectural landmark.
JOURNAL
PNAS
AUTHORS
Andreas Zembrzycki, Carlos G. Perez-Garcia, Chia-Fang Wang, Shen-Ju Chou, Dennis D.M. O’Leary
Office of Communications
Tel: (858) 453-4100
press@salk.edu