Professor
Molecular Neurobiology Laboratory
Frederick W. and Joanna J. Mitchell Chair

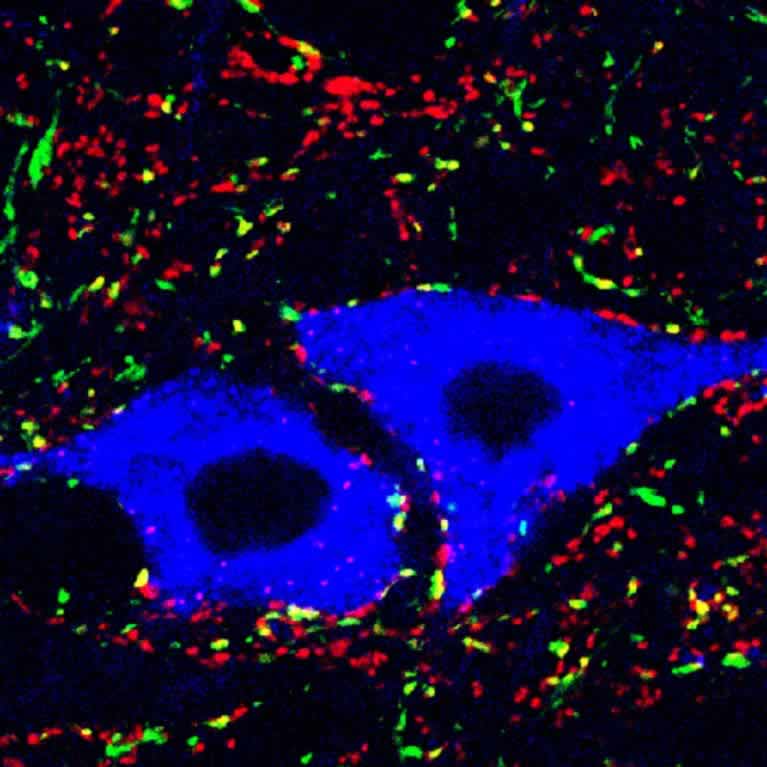
Movement is a core feature of all animal behavior, from simple behaviors such as locomotion and feeding to more complex tasks of speaking and playing a musical instrument. The control center for movement is the spinal cord, which harbors the neurons that transmit sensory information from the body to the brain. These neurons play key roles in voluntary movement and balance, and in protective motor behaviors such as withdrawing your hand from a hot object or scratching your skin to remove harmful parasites. An in-depth knowledge of how neurons in the spinal cord are connected and function is key to understanding how spinal cord injury or diseases such as ALS and Parkinson’s disrupt the ability to move and maintain balance. It is also necessary for devising therapies that restore the ability to walk after injury, and will guide the development of better therapies for chronic itch and pain, which currently rely heavily on opiate-based treatments, with the attendant issues of addiction.
The Goulding lab has established a comprehensive genetic toolkit and set of sophisticated behavioral tests that allows them to functionally dissect the circuits in the spinal cord that process sensory information and generate coordinated body movements. Over the past 15 years, Goulding has identified and characterized many of the core interneuron cell types that are required for locomotion, and has delved into the genetics and development of these neurons. Neurons called V0 neurons, he discovered, mediate alternating stepping during walking, while two classes of inhibitory neurons (V1 and V2b) control the alternating patterns of flexor and extensor muscle activity that underlies all movements of the limbs and digits. A knowledge of which neurons are needed for walking and balance can help scientists develop approaches to restore walking in people with spinal cord injuries as well as prevent falls in the elderly and people with Parkinson’s. Goulding has also leveraged his expertise in spinal neural circuitry to define the pathways that transmit and gate itch and pain, providing a better understanding of the cellular changes that underlie chronic pain and itch.
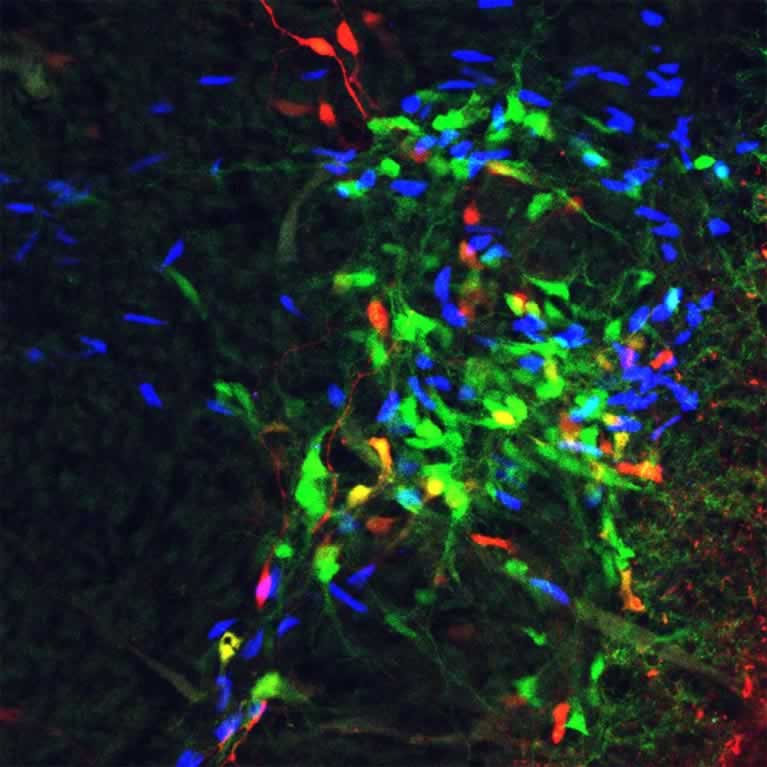
Goulding’s lab identified an important neural mechanism in the spinal cord that appears capable of sending erroneous pain signals to the brain. By charting the spinal circuits that process and transmit pain signals in mice, the study lays the groundwork for identifying ways to treat pain disorders that have no clear physical cause.
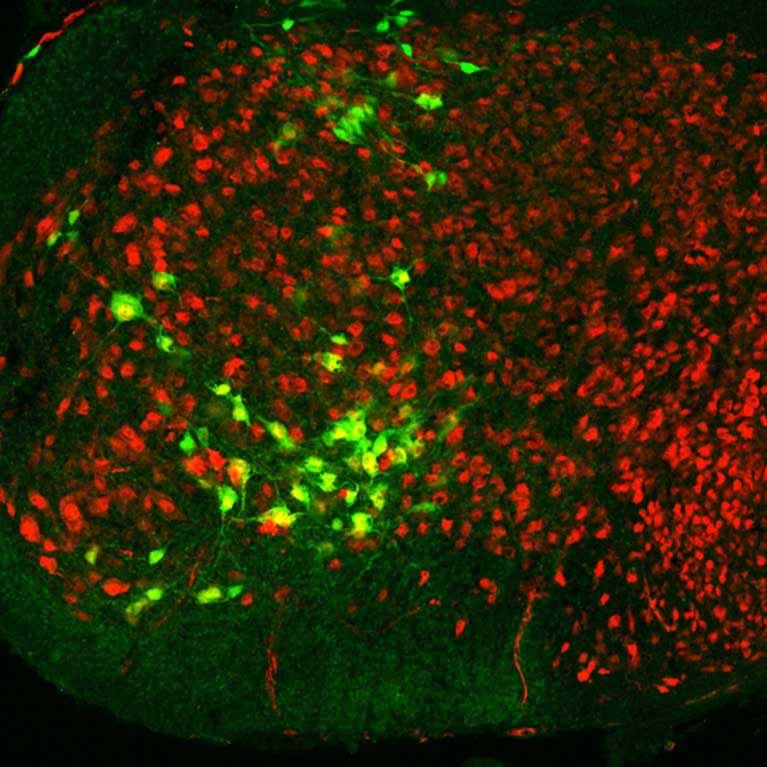
Goulding’s team mapped the neural circuitry of the spinal cord that processes the sense of light touch. A better understanding of these circuits should eventually aid in developing therapies for spinal cord injury and diseases that affect motor skills and balance, as well as the means to prevent falls in the elderly.
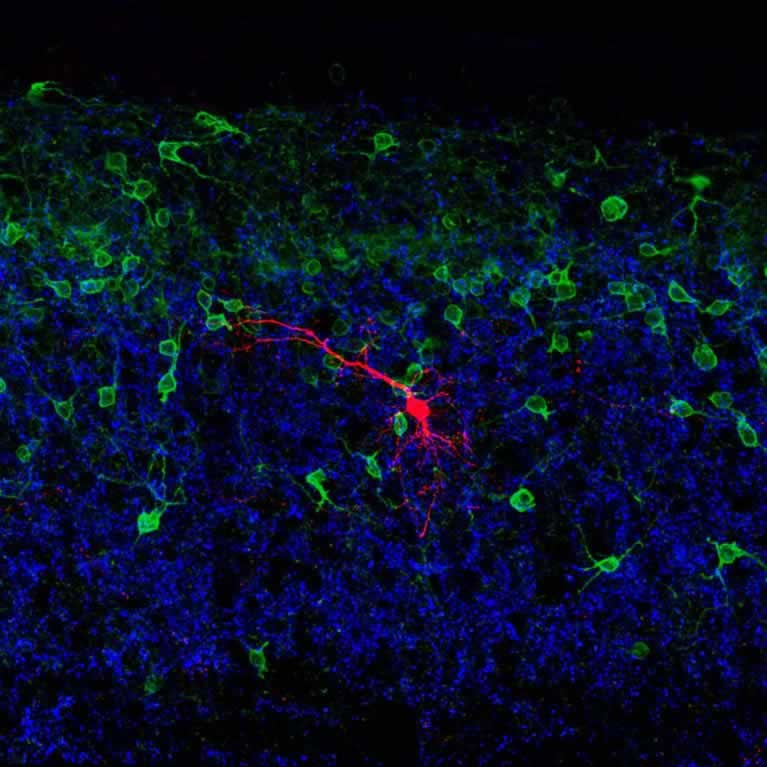
Goulding’s lab has delineated the role that multiple interneuron cell types play in controlling locomotion.
MS (Honors) Cell Biology, University of Auckland, Nz
PhD, Cellular and Molecular Biology, University of Auckland, Nz
Postdoctoral Fellow, Max Planck Institute for Biophysical Chemistry, Germany