
2023年11月28日
Salk researchers discover protein Mitf mediates the repair function in the peripheral nervous systems of mice, offering potential new therapeutic target
Salk researchers discover protein Mitf mediates the repair function in the peripheral nervous systems of mice, offering potential new therapeutic target
LA JOLLA—Each year in the United States there are more than 3 million cases of peripheral neuropathy, wherein nerves outside of the brain and spinal cord are damaged and cause pain and loss of feeling in the affected areas. Peripheral neuropathy can occur from diabetes, injury, genetically inherited disease, infection, and more. Salk scientists have now uncovered in mice a mechanism for repairing damaged nerves during peripheral neuropathy. They discovered that the protein Mitf helps turn on the repair function of specialized nervous system Schwann cells.
研究发现,发表于 细胞报告 on November 28, 2023, have the potential to inspire novel therapeutics that bolster repair function and heal peripheral neuropathy.
“We wanted to know what mechanisms control damage response in peripheral nerves under varying conditions—like acute trauma, genetic disorders, or degenerative diseases,” says senior author Professor Samuel Pfaff. “We found that Schwann cells, which are special cells in nerves that protect and support neurons’ axons, enter their repair state because of a pathway mediated by the protein Mitf.”
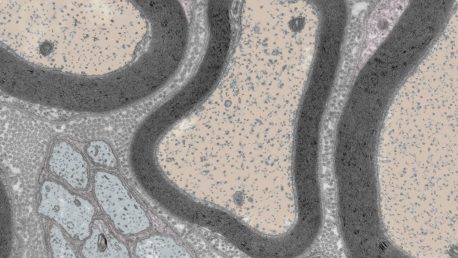
The peripheral nervous system is made up of all the nerves that branch out from the brain and spinal cord to give us sensation throughout our bodies. There are many cell types in peripheral nerves, but Pfaff and his team focus on understanding neurons, which transmit information throughout the nervous system, and Schwann cells, which protect healthy neurons and repair damaged ones.
The peripheral nervous system’s ability to repair damage is remarkable considering that the central nervous system—made up of the brain and spinal cord—is not able to repair damage. Yet, the mechanisms that orchestrate this feat have remained poorly understood.
To unravel how Schwann cells differentiate to begin repairing peripheral nerve damage, the researchers looked at mouse models of Charcot Marie Tooth disease (CMT), a type of hereditary neuropathy.
“Going into this project, I thought that when you have a genetic nerve degeneration disorder, cells are dying and recovery isn’t possible,” says first author Lydia Daboussi, a former postdoctoral researcher in Pfaff’s lab and current assistant professor at UC Los Angeles. “But our findings show that there are gene programs turned on by Mitf that repair some of the damage done in those chronic disease scenarios, and when you turn those programs off, disease symptoms get worse.”

In mice with CMT, the researchers noticed that the Schwann cells completing the repairs had high levels of Mitf in their nuclei—where the genetic instructions for how to be a Schwann cell and how to conduct repairs are stored.
Upon investigation of this relationship between Mitf and Schwann cells, they found that Mitf was in the cytoplasm of Schwann cells until sensing neuronal damage. Damage then prompted Mitf to relocate from the cytoplasm of the cell to the nucleus, where it would direct the Schwann cell to make repairs.
To validate the importance of Mitf in creating repair Schwann cells, the researchers removed Mitf altogether. In cases of both trauma and CMT, nerve repair was arrested in the absence of Mitf—demonstrating that Mitf is required for peripheral nerve repair and regeneration.
According to Daboussi, Mitf acts like a fire extinguisher. Always there, sitting in the Schwann cell, unnoticed until damage occurs. And when that damage occurs, Mitf is ready to go and immediately turns on the cell’s repair functions.
Most surprising, noted Pfaff, was that Mitf was orchestrating these repairs during a chronic disease like CMT.
“Harnessing Schwann cell repair programs has great potential in treating chronic diseases,” says Pfaff, also the Benjamin H. Lewis Chair at Salk. “It’s possible that with targeted therapeutics, we can prompt more Schwann cells to repair peripheral nerve damage and push those repairs to completion in chronic cases. Furthermore, now that we have a better grasp on the repair mechanisms, we can see if it’s possible to initiate repairs in the brain stem and spinal cord, too.”
In the future, the researchers want to look more specifically at diabetes neuropathy—the most common peripheral neuropathy condition. They also hope to explore therapeutics that bolster this repair pathway to create more Schwann cells programmed to repair damage, regardless of if the source is trauma, genetics, or development over time.
Other authors include Giancarlo Costaguta, Miriam Gullo, Nicole Jasinski, Veronica Pessino, Brendan O’Leary, Karen Lettieri, and Shawn Driscoll of Salk.
The work was supported by the Sol Goldman Charitable Trust, Howard Hughes Medical Institute, National Institutes of Health (grants NCI CCSG: P30 014195, NCI CCSG: P30 014195, S10 OD023427, S10 OD026929, 1 RO1 NS123160-01), a George E. Hewitt Fellowship, a Salk Women & Science Fellowship, and a Jonas Salk Fellowship.
DOI: https://doi.org/10.1016/j.celrep.2023.113282
日记
细胞报告
作者
Lydia Daboussi, Giancarlo Costaguta, Miriam Gullo, Nicole Jasinski, Veronica Pessino, Brendan O’Leary, Karen Lettieri, Shawn Driscoll, Samuel L. Pfaff
宣传办公室
电话:(858) 453-4100
press@salk.edu
萨尔克研究所是一个独立的非营利性研究机构,由首个安全有效的脊髓灰质炎疫苗的研发者乔纳斯·索尔克于1960年创立。该研究所的使命是推动以合作、敢于冒险为特点的基础性研究,以应对癌症、阿尔茨海默病和农业脆弱性等社会最紧迫的挑战。这项基础科学支撑着所有的转化研究,产生有助于全球新药和创新的见解。.