
July 30, 2019
Salk scientists uncover a mechanism linking a protein mutation with abnormal nervous system development
Salk scientists uncover a mechanism linking a protein mutation with abnormal nervous system development
LA JOLLA—Neurodevelopmental disorders arising from rare genetic mutations can cause atypical cognitive function, intellectual disability, and developmental delays, yet it is unclear why and how this happens. Scientists suspected a mutation in a complex of proteins could be the culprit for a group of rare genetic disorders and, now, Salk Institute researchers have identified the molecular mechanism linking this mutation with abnormal nervous system development. The team’s findings, published in Molecular Cell on July 30, 2019, bring researchers one step closer to understanding neurodevelopmental disorders, such as Nicolaides-Baraitser syndrome and others.
“For the first time, we have been able to characterize the mechanism of a known gene mutation implicated in neurodevelopmental disorders,” says Assistant Professor 戴安娜·哈格里夫斯, senior author and holder of the Richard Heyman and Anne Daigle Endowed Developmental Chair.
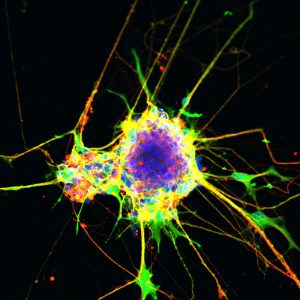
点击此处 用于高分辨率图像。.
版权:萨克研究所
The root cause has to do with a complex of proteins called the SWI/SNF complex, which is involved in DNA regulation, and, when mutated, is associated with Nicolaides-Baraitser syndrome, Coffin-Siris syndrome, autism and even some cancers. These complexes repackage and reshape the DNA in the nucleus to either enable or prevent access to genes. And yet, scientists do not know how mutations in individual subunits of the SWI/SNF complex affect its function.
“We sought to understand how a single mutation in the SMARCA2 subunit affected brain development,” says Fangjian Gao, the paper’s first author and a postdoctoral fellow at Salk. “We expected to see some effect on the neurodevelopmental pathways, but we were not sure how, specifically.”

点击此处 用于高分辨率图像。.
版权:萨克研究所
The scientists turned to cell cultures in a dish to model growth patterns in afflicted brain cells versus normal brain cells. They used the gene-editing technique CRISPR to mimic the SMARCA2 mutation observed in Nicolaides-Baraitser syndrome. Notably, the researchers found that healthy cells had minimal SMARCA2 activity. The cells with the mutation, however, had a dramatic increase in SMARCA2 activity and a significantly impaired ability to generate precursors to neurons, called neural progenitor cells. In this highly activated state, SMARCA2 affected the function of the normal SWI/SNF complex. This led to a domino effect with changes in gene expression resulting in abnormal brain development.
“By better understanding this mutation in SMARCA2, we have tapped into what looks like a core developmental process that could be perturbed in disease states such as autism or even cancer,” says Hargreaves.
Other authors included Nicholas J. Elliott, Josephine Ho and Maxim N. Shokhirev of Salk, along with Alexzander Sharp of the University of California San Diego.
The work was funded by the Helmsley Trust, the National Institutes of Health (9R35 GM128943-01 and R00 CA184043-03) and the V Foundation for Cancer Research (V2016-006).
DOI: 10.1016/j.molcel.2019.06.024
日记
Molecular Cell
标题
Heterozygous mutations in SMARCA2 reprogram the enhancer landscape by global retargeting of SMARCA4
作者
Fangjian Gao, Nicholas J. Elliott, Josephine Ho, Alexzander Sharp, Maxim N. Shokhirev and Diana C. Hargreaves
期刊封面

宣传办公室
电话:(858) 453-4100
press@salk.edu
萨尔克研究所是一个独立的非营利性研究机构,由首个安全有效的脊髓灰质炎疫苗的研发者乔纳斯·索尔克于1960年创立。该研究所的使命是推动以合作、敢于冒险为特点的基础性研究,以应对癌症、阿尔茨海默病和农业脆弱性等社会最紧迫的挑战。这项基础科学支撑着所有的转化研究,产生有助于全球新药和创新的见解。.