
June 30, 2015
Salk scientists find that dietary fat, coupled with a natural hormone, can relieve metabolic dysfunction associated with mitochondrial disease in mice
Salk scientists find that dietary fat, coupled with a natural hormone, can relieve metabolic dysfunction associated with mitochondrial disease in mice
LA JOLLA–Mice that have a genetic version of mitochondrial disease can easily be mistaken for much older animals by the time they are nine months old: they have thinning grey hair, osteoporosis, poor hearing, infertility, heart problems and have lost weight. Despite having this disease at birth, these mice have a “secret weapon” in their youth that staves off signs of aging for a time.
New research from the Salk Institute reveals how a longevity hormone helps these mice–born with thousands of mutations in their energy-generating mitochondria–maintain metabolic homeostasis at a young age. This work provides insight into potential new therapies for mitochondrial and metabolic diseases in humans.
“These findings help us understand the link between diet, health and aging, and they give us the potential to dissect these connections in a molecular way that could lead to therapeutics,” says 罗纳德·埃文斯, director of Salk’s 基因表达实验室 and senior author of the new paper, published June 29, 2015 in 美国国家科学院院刊.
When Evans and collaborators studied these mice, they found that the longevity-promoting endocrine hormone FGF21 was highly active, even though these mice display hallmarks of accelerated aging.
“FGF21 is classically thought of as an anti-aging gene, turned on by exercise or fasting and suspected to extend lifespan,” says Christopher Wall, a graduate student in the Evans lab at the time of the study and first author of the new paper. “Yet these mice ultimately age prematurely, despite having high levels of this hormone.”
To explain this paradox, the team found that FGF21 actually assists stressed mitochondria and reprograms the metabolic state of these mice. In the paper, Evans and collaborators describe how FGF21 appears to switch tissues with important metabolic functions from burning sugar to burning fat–which is easier fuel for the dysfunctional mitochondria to process.
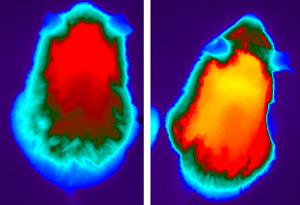
In young but rapidly aging mice, high-fat diet feeding (right) ramps up heat production and metabolic activity relative to cooler mice fed a normal chow diet (left).
点击此处 用于高分辨率图像。.
图片:由萨克生物研究所提供
“This works for a while in these mice, but eventually their bodies run out of fat to burn,” says Michael Downes, a senior staff scientist at Salk involved in the new study. “FGF21 needs some additional fat from the diet to keep working.” The team also found that feeding the mice a high-fat diet amplifies the effects of FGF21, granting them complete resistance to diet-induced obesity and many of its associated diseases.
“What we believe now is that turning on FGF21 is an adaptation to help this animal maintain its metabolic health,” says Evans, a Howard Hughes Medical Institute investigator and the March of Dimes Chair in Molecular and Developmental Biology. “If the mice didn’t have this hormonal ‘fountain of youth,’ they might age even more quickly than they already do.”
The new observation has the potential to impact human health in multiple ways, says Evans. Elevated FGF21 levels have previously been observed in people with mitochondrial diseases, so increasing dietary fat intake could potentially ease their symptoms by helping the hormone reduce the strain on the mitochondria. At the same time, these findings suggest that using drugs to mildly stress mitochondria could activate a beneficial FGF21-dependent metabolic shift in patients suffering from obesity and other metabolic diseases.
“We want to look at the interaction between FGF21 and dietary fat in human patients with mitochondrial disease now,” says Downes, “and start to see how we can manipulate this dynamic in different ways.”
Other researchers on the study were Jamie Whyte, Jae M. Suh, Weiwei Fan, Brett Collins, Ruth Yu, and Annette R. Atkins of the Salk Institute; Christopher Liddle of the University of Sydney; and Jane C. Naviaux, Kefeng Li, Andrew Taylor Bright, William A. Alaynick, and Robert K. Naviaux of the University of California, San Diego School of Medicine.
该研究得到了 霍华德·休斯医学研究所, , 那个 美国国立卫生研究院, , 那个 格伦医学研究基金会, , 那个 Leona M. 和 Harry B. Helmsley 慈善信托基金, 益普森/生物测量, , 那个 埃里森医学基金会, , 那个 塞缪尔·瓦克斯曼癌症研究所, the Jane Botsford Johnson Foundation, the University of California, San Diego (UCSD) Christini Foundation, the UCSD Mitochondrial Research Fund, ,和 Wright Family Foundation.
日记
美国国家科学院院刊
作者
Christopher E. Wall, Jamie Whyte, Jae M. Suh, Weiwei Fan, Brett Collins, Ruth Yu, Annette R. Atkins, Michael Downes, and Ronald M. Evans of the Salk Institute; Christopher Liddle of the University of Sydney; Jane C. Naviaux, Kefeng Li, Andrew Taylor Bright, William A. Alaynick, and Robert K. Naviaux of the University of California at San Diego School of Medicine
宣传办公室
电话:(858) 453-4100
press@salk.edu
萨尔克研究所是一个独立的非营利性研究机构,由首个安全有效的脊髓灰质炎疫苗的研发者乔纳斯·索尔克于1960年创立。该研究所的使命是推动以合作、敢于冒险为特点的基础性研究,以应对癌症、阿尔茨海默病和农业脆弱性等社会最紧迫的挑战。这项基础科学支撑着所有的转化研究,产生有助于全球新药和创新的见解。.