
2017年12月7日
Approach could also be applied to reversing aging and age-related diseases such as hearing loss and macular degeneration
Approach could also be applied to reversing aging and age-related diseases such as hearing loss and macular degeneration
拉霍亚—索尔克研究所的科学家们创建了CRISPR/Cas9基因组编辑技术的一个新版本,该版本使他们能够在不破坏DNA的情况下激活基因,这可能克服使用基因编辑技术治疗人类疾病的一个主要障碍。.
Most CRISPR/Cas9 systems work by creating “double-strand breaks” (DSBs) in regions of the genome targeted for editing or for deletion, but many researchers are opposed to creating such breaks in the DNA of living humans. As a proof of concept, the Salk group used their new approach to treat several diseases, including diabetes, acute kidney disease, and muscular dystrophy, in mouse models.
“Although many studies have demonstrated that CRISPR/Cas9 can be applied as a powerful tool for gene therapy, there are growing concerns regarding unwanted mutations generated by the double-strand breaks through this technology,” says 胡安·卡洛斯·伊兹皮苏亚·贝尔蒙特, a professor in Salk’s Gene Expression Laboratory and senior author of the new paper, published in 细胞 on December 7, 2017. “We were able to get around that concern.”
点击此处 用于高分辨率图像。.
版权:萨克研究所
In the original CRISPR/Cas9 system, the enzyme Cas9 is coupled with guide RNAs that target it to the right spot in the genome to create DSBs. Recently, some researchers have started using a “dead” form of Cas9 (dCas9), which can still target specific places in the genome, but no longer cuts DNA. Instead, dCas9 has been coupled with transcriptional activation domains—molecular switches—that turn on targeted genes. But the resulting protein—dCas9 attached to the activator switches—is too large and bulky to fit into the vehicle typically used to deliver these kinds of therapies to cells in living organisms, namely adeno-associated viruses (AAVs). The lack of an efficient delivery system makes it very difficult to use this tool in clinical applications.
Izpisua Belmonte’s team combined Cas9/dCas9 with a range of different activator switches to uncover a combination that worked even when the proteins were not fused to one another. In other words, Cas9 or dCas9 was packaged into one AAV, and the switches and guide RNAs were packaged into another. They also optimized the guide RNAs to make sure all the pieces ended up at the desired place in the genome, and that the targeted gene was strongly activated.
“The components all work together in the organism to influence endogenous genes,” says Hsin-Kai (Ken) Liao, a staff researcher in the Izpisua Belmonte lab and co–first author of the new paper. In this way, the technology operates epigenetically, meaning it influences gene activity without changing the DNA sequence.

点击此处 用于高分辨率图像。.
版权:萨克研究所
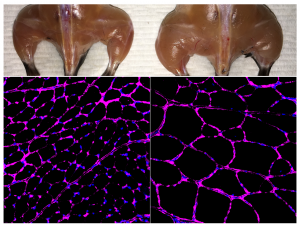
To test the method, the researchers used mouse models of acute kidney injury, type 1 diabetes and a form of muscular dystrophy. In each case, they engineered their CRISPR/Cas9 system to boost the expression of an endogenous gene that could potentially reverse disease symptoms. In the case of kidney disease, they activated two genes known to be involved in kidney function, and observed not only increased levels of the proteins associated with those genes, but improved kidney function following an acute injury. For type 1 diabetes, they aimed to boost the activity of genes that could generate insulin-producing cells. Once again, the treatment worked, lowering blood glucose levels in a mouse model of diabetes. For muscular dystrophy, the researchers expressed genes that have been previously shown to reverse disease symptoms, including one particularly large gene that cannot easily be delivered via traditional virus-mediated gene therapies.
“We were very excited when we saw the results in mice,” adds Fumiyuki Hatanaka, a research associate in the lab and co–first author of the paper. “We can induce gene activation and at the same time see physiological changes.”
Izpisua Belmonte’s team is now working to improve the specificity of their system and to apply it to more cell types and organs to treat a wider range of human diseases, as well as to rejuvenate specific organs and to reverse the aging process and age-related conditions such as hearing loss and macular degeneration. More safety tests will be needed before human trials, they say.
Other researchers on the study were Toshikazu Araoka, Pradeep Reddy, Min-Zu Wu, Takayoshi Yamauchi, Masahiro Sakurai, David O’Keefe, and Concepcion Rodriguez Esteban of the Salk Institute; Yinghui Sui, Cheng-Jang Wu, and Li-Fan Lu of the University of California, San Diego; Estrella Nuñez of Universidad Católica; Pedro Guillén of Fundación Pedro Guillén; and Josep Campistol of Hospital Clinic of Barcelona.
The work and the researchers involved were supported by grants from the Uehara Memorial Foundation, the Moxie Foundation, The Leona M. and Harry B. Helmsley Charitable Trust, the G. Harold and Leila Y. Mathers Charitable Foundation, Fundación Dr. Pedro Guillén, Asociación de Futbolistas Españoles (AFE), Universidad Católica de San Antonio de Murcia (UCAM), the National Institutes of Health, Howard Hughes Medical Institute, and a Calouste Gulbenkkian Foundation Fellowship.
日记
细胞
作者
Hsin-Kai Liao, Fumiyuki Hatanaka, Toshikazu Araoka, Pradeep Reddy, Min-Zu Wu, Yinghui Sui, Takayoshi Yamauchi, Masahiro Sakurai, David O'Keefe, Cheng-Jang Wu, Li-Fan Lu, Estrella Nuñez, Pedro Guillén, Josep M. Campistol, Concepcion Rodriguez Esteban, Juan Carlos Izpisua Belmonte
宣传办公室
电话:(858) 453-4100
press@salk.edu
萨尔克研究所是一个独立的非营利性研究机构,由首个安全有效的脊髓灰质炎疫苗的研发者乔纳斯·索尔克于1960年创立。该研究所的使命是推动以合作、敢于冒险为特点的基础性研究,以应对癌症、阿尔茨海默病和农业脆弱性等社会最紧迫的挑战。这项基础科学支撑着所有的转化研究,产生有助于全球新药和创新的见解。.