
December 7, 2010
LA JOLLA, CA—Better known as the light sensor that sets the body’s biological clock, melanopsin also plays an important role in vision: Via its messengers-so-called melanopsin-expressing retinal ganglion cells, or mRGCs-it forwards information about the brightness of incoming light directly to conventional visual centers in the brain, reports an international collaboration of scientists in this week’s issue of PLoS生物学.
The findings reveal a new role for mRGCs during image-forming vision and suggest that these cells could make a significant contribution to assessing the intensity of light and supporting vision even in people with advanced retinal degeneration, the researchers say.
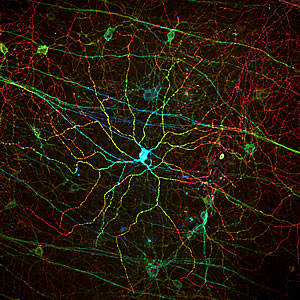
Melanopsin, a light receptor that measures the intensity of incoming light is found in rare, specialized cells embedded in the retina. The different colors corresponds to different depths of the imaged tissue sample.
Image: Courtesy of Dr. Satchin Panda, Salk Institute for Biological Studies and Dr. James Fitzpatrick, Waitt Advanced Biophotonics Center
“Millions of people worldwide suffer varying degrees of blindness because of rod and cone degeneration or dysfunction, but many of them can still perceive differences in brightness,” says senior author Satchidananda (Satchin) Panda, Ph.D., an assistant professor in the Regulatory Biology Laboratory at the Salk Institute for Biological Studies. “Melanopsin-expressing RGCs typically survive even complete rod and cone loss and could explain the light responses under these conditions,” he adds.
For the greater part of 100 years, it was thought that the ability to convert light into electrical signals in the mammalian retina was restricted to only two types of photoreceptors: rods and cones. This view changed dramatically when Panda discovered the existence of a third type of mammalian photoreceptor, which is only present in a few thousand cells embedded in the deeper layers of the retina.
Melanopsin, a photopigment that measures the intensity of incoming light, is fundamentally different from the classical rod and cone opsins, which help us see. For one, it is much less sensitive to light and has far less spatial resolution-characteristics that fit perfectly with this light sensor’s primary function of signaling changes in ambient light levels to the brain throughout the day.
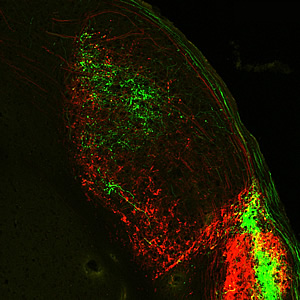
Melanopsin-expressing retinal ganglion cells extend their axons-long, slender projections that connect with other neurons-all the way to the primary processing center for visual information received from the brain.
Image: Courtesy of Dr. Satchin Panda, Salk Institute for Biological Studies and Dr. James Fitzpatrick, Waitt Advanced Biophotonics Center
It sends its signals directly to the human circadian clock, which sits just above the point where the optic nerves cross. Although only half the size of a pencil eraser, it synchronizes the body’s daily rhythms with the rising and setting of the sun, telling the body when it is time to go to sleep, when to be hungry and when to wake up. But it does more than that. Just like a meter in a camera that allows the aperture to be adjusted, mRGCs also control pupil size.
Until now, however, it was unknown whether mRGCs also contribute to conventional image-forming vision, especially the as-yet poorly understood mechanism of “brightness” and “lightness” perception.
To find out, Panda and his collaborators at the University of Manchester traced individual mRGCs’ axons-long, slender projections that connect with other neurons-from the retina through the circadian clock and onward. They discovered that the axons reached all the way to the LGN, short for lateral geniculate nucleus, the primary processing center for visual information received from the retina.
“We found widespread light responses in the LGN and visual cortex, even in mice lacking functional rods and cones, which are often used as a model of advanced retinal degeneration,” says Panda, who hopes that one day it might be possible to impart vision to blind individuals by gene therapy with a re-engineered melanopsin.
“The density of mRGCs in the retina is too low for any meaningful resolution,” he says. “But if we could express melanopsin in a greater number of cells, we might be able to increase resolution to a point that allows blind people to safely navigate their environment.”
Researchers who also contributed to the work include Timothy M. Brown, John Gigg and Robert J. Lucas at the University of Manchester, Manchester, United Kingdom; Carlos Gias, Ma’ayan Semo and Peter J. Coffey at University College London, London, United Kingdom; as well as Megumi Hatori, and Sheena Rachel Keding at the Salk Institute for Biological Studies in La Jolla.
The work was funded by grants from the Wellcome Trust, the National Institutes of Health, the Dana Foundation, a Pew Scholar award and the London Project to Cure Blindness.
关于索尔克生物研究所:
索尔克生物研究所是世界顶尖的基础研究机构之一,其国际知名的教职人员在一个独特、协作和富有创造性的环境中,深入探究生命科学的基本问题。索尔克科学家们致力于发现和指导未来几代研究人员,通过研究神经科学、遗传学、细胞和植物生物学以及相关学科,在癌症、衰老、阿尔茨海默氏症、糖尿病和传染病的认识方面做出了开创性的贡献。.
学院取得了许多成就,获得了包括诺贝尔奖和美国国家科学院院士在内的无数荣誉。该研究所由脊髓灰质炎疫苗先驱 Jonas Salk 博士于 1960 年创立,是一家独立的非营利组织和建筑地标。.
索尔克研究所为基础研究领域的卓越科学成就而自豪地庆祝了五十周年。.
宣传办公室
电话:(858) 453-4100
press@salk.edu