
April 22, 2026
LA JOLLA—Fluorescent proteins have revolutionized science, enabling researchers to tag and visualize individual molecules in living cells, tissues, and animals. Using these tools, researchers have watched viruses infect cells in real time, observed cellular trash collection, and tracked the signaling that spurs tumor growth.

Salk scientists and collaborators at Albert Einstein College of Medicine have advanced this visualization technology. The new technology, called visible-spectrum antigen-stabilizable fluorescent nanobodies (VIS-Fbs), was validated in multiple mammalian cell types and provides a powerful tool for a wide range of life science research applications.
The study was published in 自然方法 on April 22, 2026.
“This work establishes a versatile platform for imaging proteins with high specificity and minimal background,” says co-corresponding author 阿克塞尔-尼默雅恩,博士, professor and Françoise Gilot-Salk Chair at Salk. “It opens new opportunities to study how molecular and cellular processes unfold in real time across diverse biological systems.”
The innovation began with tiny protein fragments called nanobodies, which can be engineered to bind specific protein targets in living cells. When fused to fluorescent proteins, these nanobody-based probes can reveal where target proteins are located and how they behave. However, conventional versions can still generate signal even when unbound, creating background fluorescence that can obscure fine details.
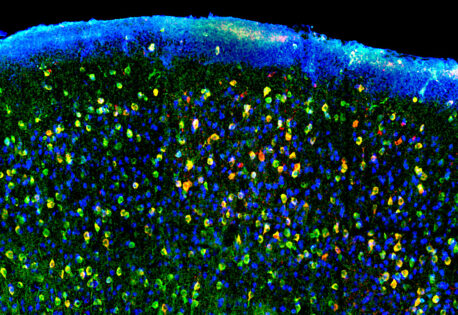
The Salk and Einstein team designed a new type of probe that retains the targeting power of nanobodies while greatly reducing background fluorescence. VIS-Fbs become stable and fluorescent only when bound to their intended target. This binding-dependent (“on-demand”) fluorescence reduces background noise by up to about a hundredfold, enabling much sharper visualization of protein location and dynamics.
In addition, the researchers developed multiple versions of this new probe that fluoresce across nearly the entire visible spectrum, from blue to far red. With this many color options, multiple cellular targets can be tracked simultaneously. Certain VIS-Fb variants can also be switched “on” and “off” with light, making it possible to follow protein behavior over time with high spatial and temporal precision. The researchers also established a modular design framework, enabling rapid adaptation of VIS-Fb probes to different targets and functional readouts.
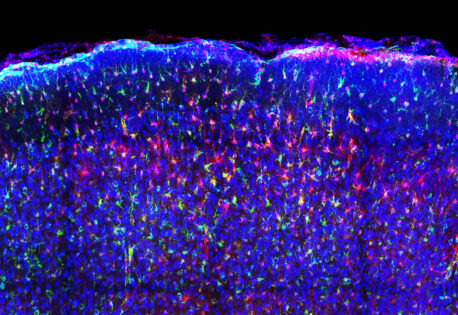
The new technology will allow scientists to gain more accurate, timely insight into cellular activity—even in complex environments like living brain tissue. The researchers demonstrated VIS-Fbs’ capabilities across a range of living models.
In mouse models, VIS-Fb probes enabled selective labeling and ratiometric imaging of calcium activity in neurons and astrocytes during behavior. In zebrafish, the technology allowed real-time tracking of dynamic changes during early development and in response to drugs that alter signaling pathways.
“Our results show that this imaging platform offers a much clearer and more precise view of how proteins behave inside living systems,” says co-corresponding author of the study Vladislav Verkhusha, PhD, professor and co-director of the Gruss Lipper Biophotonics Center at Albert Einstein College of Medicine. “It opens the door to studying complex biological processes, such as cell signaling, development, and disease progression, in new ways.”
Other authors include Erin Carey of Salk; Natalia Barykina, Juliana Mendonça-Gomes, and Sofia de Oliveira of the Albert Einstein College of Medicine; and Olena Oliinyk of the University of Helsinki.
This study was funded by the National Institutes of Health (GM122567, NS123719, GM147416), Jane and Aatos Erkko Foundation, Research Council of Finland, Finland Cancer Foundation, Chan Zuckerberg Initiative Foundation, NOMIS Foundation (Salk’s Neuroimmunology Initiative), and Edwards-Yeckel Research Foundation.
DOI: 10.1038/s41592-026-03056-3
日记
自然方法
作者
Natalia V. Barykina, Erin M. Carey, Olena S. Oliinyk, Juliana M. Mendonça-Gomes, Sofia de Oliveira, Axel Nimmerjahn, and Vladislav V. Verkhusha
宣传办公室
电话:(858) 453-4100
press@salk.edu
萨尔克研究所是一个独立的非营利性研究机构,由首个安全有效的脊髓灰质炎疫苗的研发者乔纳斯·索尔克于1960年创立。该研究所的使命是推动以合作、敢于冒险为特点的基础性研究,以应对癌症、阿尔茨海默病和农业脆弱性等社会最紧迫的挑战。这项基础科学支撑着所有的转化研究,产生有助于全球新药和创新的见解。.