
April 23, 2015
Salk scientists use molecular "scissors" to eliminate mitochondrial mutations in eggs and embryos
Salk scientists use molecular "scissors" to eliminate mitochondrial mutations in eggs and embryos
LA JOLLA–For thousands of women around the globe carrying a mitochondrial disease, having a healthy child can be a gamble. This set of diseases affect mitochondria, tiny powerhouses that generate energy in the body’s cells and are passed exclusively from mother to child.
Women wishing to prevent their children from inheriting mitochondrial diseases have typically relied on preimplantation genetic diagnosis to pick the healthiest embryos, but that is no guarantee of having a healthy baby.
Now, researchers at Salk Institute have developed a simple technique to eliminate mitochondrial mutations from eggs or early embryos, which has the potential to prevent babies from inheriting mitochondrial diseases. Their approach is described in the April 23, 2015 issue of 细胞.
“Currently, there are no treatments for mitochondrial diseases,” says senior author 胡安·卡洛斯·伊兹皮苏亚·贝尔蒙特, professor in Salk’s 基因表达实验室 and holder of the Roger Guillemin Chair. “Our technology may offer new hope for mitochondrial disease carriers wishing to have children without the disease.”
Living cells can have hundreds–or even thousands–of mitochondria, each of which contains their own DNA, a small collection of 37 genes that are essential for the organelle’s function. Mutations in these crucial genes can cause a wide range of diseases and can lead to fatality at birth, a life expectancy of only a few years or devastating symptoms for decades.
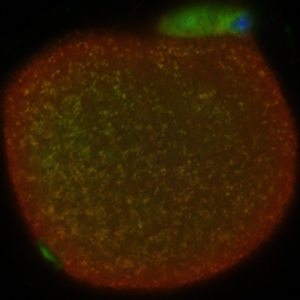
Salk Institute researchers used specialized molecules (called nucleases) to eliminate mutated DNA from mitochondria. This image shows the location of nucleases with mitochondria (yellow) in the egg.
点击此处 用于高分辨率图像。.
图片:由萨克生物研究所提供
“Most current approaches are trying to develop treatments for patients who are already suffering from these diseases,” says Alejandro Ocampo, a research associate in Izpisua Belmonte’s lab and one of the first authors of the paper. “Instead, we thought of preventing the transmission of these mutations early in development.”
Izpisua Belmonte and his colleagues turned to two types of molecules: restriction endonucleases and transcription activator-like effector nucleases (TALENs). These nucleases can be engineered to cut specific strands of DNA, functioning as a type of molecular “scissors.” The Salk team designed nucleases to cut only mitochondrial DNA that contained specific, disease-causing mutations in eggs or embryos, leaving healthy mitochondria intact.
“We might not be able to eliminate one hundred percent of the mutated copies of mitochondrial DNA,” says Pradeep Reddy, another research associate in the Izpisua Belmonte lab and first author of the new paper. “But you don’t need to eliminate all of the mutated copies: just reducing the percentage significantly enough can prevent the disease in the next generation.”
As proof of concept, the scientists, using mice containing two types of mitochondrial DNA, selectively prevented the transmission of one of the types to the next generation using specific nucleases in both eggs and one-cell embryos. Baby mice generated by this approach developed normally to adulthood. In addition, this method let the researchers successfully reduce the levels of mutated mitochondrial DNA responsible for two human mitochondrial diseases.
In the United Kingdom, lawmakers have recently approved the use of another technology known as mitochondrial replacement to prevent the transmission of mitochondrial diseases. This technology is based on the transfer of the nuclear genome from one patient’s embryo into donor embryos with healthy mitochondria. “The clinical application of our technique does not require donor eggs,” says Ocampo. “We are just performing a single injection into the patient’s egg or one-cell embryo, which is technically easier than mitochondrial replacement.”
Izpisua Belmonte and colleagues are now investigating the possibility of translating this technology to the clinic in human eggs and embryos.
Other researchers on the study were Keiichiro Suzuki, Jinping Luo, Atsushi Sugawara, Daiji Okamura, Jun Wu, David Lam, Concepcion Rodriguez Esteban, and Ignacio Sancho-Martinez of the Salk Institute; Sandra R. Bacman, Sion L. Williams, and Carlos T. Moraes of the University of Miami; Yuji Tsunekawa of the RIKEN Center for Developmental Biology; Xiong Xiong and Huimin Zhao of the University of Illinois at Urbana-Champaign; Nuria Montserrat of the Institute for Bioengineering of Catalonia; Guang-Hui Liu of the Chinese Academy of Sciences; Dolors Manau, Salva Civico, Francesc Cardellach and Josep Maria Campistol of the University of Barcelona; and Maria del Mar O’Callaghan and Jaime Campistol of the Hospital Sant Joan de Déu Barcelona.
这项工作得到了...的支持 Leona M. 和 Harry B. Helmsley 慈善信托基金, , 那个 美国国立卫生研究院, , 那个 National Basic Research Program of China, the Strategic Priority Research Program of the Chinese Academy of Sciences, the National Natural Science Foundation of China, , 那个 JDM Fund, , 那个 Muscular Dystrophy Association, , 那个 United Mitochondrial Disease Foundation, , 那个 Florida Department of Health 和 G.哈罗德和莱拉·Y·马瑟斯慈善基金会.
关于索尔克生物研究所:
索尔克生物学研究所是世界顶尖的基础研究机构之一,其国际知名的教职员工在一个独特的、合作和富有创造力的环境中,探索生命科学的基本问题。索尔克科学家致力于科学发现和培养下一代研究人员,通过对神经科学、遗传学、细胞和植物生物学以及相关学科的研究,在癌症、衰老、阿尔茨海默病、糖尿病和传染病等领域取得了开创性的贡献,加深了我们对这些疾病的理解。.
教职员工的成就获得了无数荣誉,包括诺贝尔奖和美国国家科学院院士资格。该研究所由脊髓灰质炎疫苗先驱 Jonas Salk 医生于 1960 年创立,是一家独立的非营利组织和建筑地标。.
日记
细胞
作者
Pradeep Reddy, Alejandro Ocampo, Keiichiro Suzuki, Jinping Luo, Atsushi Sugawara, Daiji Okamura, Jun Wu, David Lam, Concepcion Rodriguez Esteban, Ignacio Sancho-Martinez, and Juan Carlos Izpisua Belmonte of the Salk Institute; Sandra R. Bacman, Sion L. Williams, and Carlos T. Moraes of the University of Miami; Yuji Tsunekawa of the RIKEN Center for Developmental Biology; Xiong Xiong and Huimin Zhao of the University of Illinois at Urbana-Champaign; Nuria Montserrat of the Institute for Bioengineering of Catalonia; Guang-Hui Liu of the Chinese Academy of Sciences; Dolors Manau, Salva Civico, Francesc Cardellach and Josep Maria Campistol of the University of Barcelona; and Maria del Mar O’Callaghan and Jaime Campistol of the Hospital Universitari Sant Joan de Deu
宣传办公室
电话:(858) 453-4100
press@salk.edu