
2017 年 2 月 14 日
Salk scientists create synthetic brain systems called “circuitoids” to better understand dysfunctional movements in Parkinson’s, ALS and other diseases
Salk scientists create synthetic brain systems called “circuitoids” to better understand dysfunctional movements in Parkinson’s, ALS and other diseases
LA JOLLA—Not everyone is Fred Astaire or Michael Jackson, but even those of us who seem to have two left feet have got rhythm—in our brains. From breathing to walking to chewing, our days are filled with repetitive actions that depend on the rhythmic firing of neurons. Yet the neural circuitry underpinning such seemingly ordinary behaviors is not fully understood, even though better insights could lead to new therapies for disorders such as Parkinson's disease, ALS and autism.
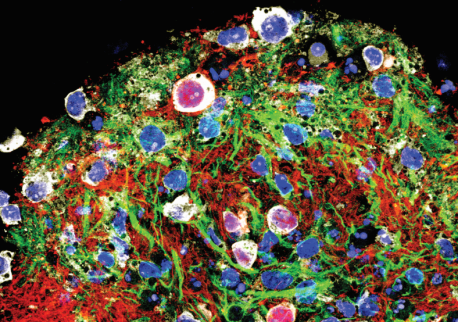
Recently, neuroscientists at the Salk Institute used stem cells to generate diverse networks of self-contained spinal cord systems in a dish, dubbed circuitoids, to study this rhythmic pattern in neurons. The work, which appears online in the February 14, 2017, issue of eLife, reveals that some of the circuitoids—with no external prompting—exhibited spontaneous, coordinated rhythmic activity of the kind known to drive repetitive movements.
"It's still very difficult to contemplate how large groups of neurons with literally billions if not trillions of connections take information and process it," says the work's senior author, Salk Professor Samuel Pfaff, who is also a 霍华德·休斯医学研究所 investigator and holds the Benjamin H. Lewis Chair. "But we think that developing this kind of simple circuitry in a dish will allow us to extract some of the principles of how real brain circuits operate. With that basic information maybe we can begin to understand how things go awry in disease."
点击此处 用于高分辨率图像。.
版权:萨克研究所
Nerve cells in your brain and spinal cord connect to one another much like electronic circuits. And just as electronic circuits consist of many components, the nervous system contains a dizzying array of neurons, often resulting in networks with many hundreds of thousands of cells. To model these complex neural circuits, the Pfaff lab prompted embryonic stem cells from mice to grow into clusters of spinal cord neurons, which they named circuitoids. Each circuitoid typically contained 50,000 cells in clumps just large enough to see with the naked eye, and with different ratios of neuronal subtypes.
With molecular tools, the researchers tagged four key subtypes of both excitatory (promoting an electrical signal) and inhibitory (stopping an electrical signal) neurons vital to movement, called V1, V2a, V3 and motor neurons. Observing the cells in the circuitoids in real time using high-tech microscopy, the team discovered that circuitoids composed only of V2a or V3 excitatory neurons or excitatory motor neurons (which control muscles) spontaneously fired rhythmically, but that circuitoids comprising only inhibitory neurons did not. Interestingly, adding inhibitory neurons to V3 excitatory circuitoids sped up the firing rate, while adding them to motor circuitoids caused the neurons to form sub-networks, smaller independent circuits of neural activity within a circuitoid.
“These results suggest that varying the ratios of excitatory to inhibitory neurons within networks may be a way that real brains create complex but flexible circuits to govern rhythmic activity,” says Pfaff. “Circuitoids can reveal the foundation for complex neural controls that lead to much more elaborate types of behaviors as we move through our world in a seamless kind of way.”

点击此处 用于高分辨率图像。.
版权:萨克研究所
Because these circuitoids contain neurons that are actively functioning as an interconnected network to produce patterned firing, Pfaff believes that they will more closely model a normal aspect of the brain than other kinds of cell culture systems. Aside from more accurately studying disease processes that affect circuitry, the new technique also suggests a mechanism by which dysfunctional brain activity could be treated by altering the ratios of cell types in circuits.
Other authors included: Matthew J. Sternfeld, Christopher A. Hinckley, Niall J. Moore, Matthew T. Pankratz, Kathryn L. Hilde, Shawn P. Driscoll, Marito Hayashi, Neal D. Amin, Dario Bonanomi, Wesley D. Gifford, and Martyn Goulding of Salk; and Kamal Sharma of the University of Illinois, Chicago.
这项工作由...资助 国家癌症研究所 at the National Institutes of Health; the 罗斯希尔斯基金会; the H. A. and Mary K. Chapman Charitable Trust; the 加州大学圣地亚哥分校, Neurosciences Graduate Program; a U.S. National Research Service Award fellowship from the U.S. National Institutes of Health National Institute of Neurological Disorders and Stroke; the 国家科学基金会; the Japanese Ministry of Education, Culture, Sports, Science, and Technology Long-Term Student Support Program; the Timken-Sturgis Foundation; the 加州再生医学研究所; the Howard Hughes Medical Institute; the 克里斯托弗和达纳·里夫基金会; the Marshall Heritage Foundation; and the Sol Goldman Charitable Trust.
日记
eLife
作者
Matthew J Sternfeld, Christopher A. Hinckley, Niall J. Moore, Matthew T. Pankratz, Kathryn L. Hilde, Shawn P Driscoll, Marito Hayashi, Neal D. Amin, Dario Bonanomi, Wesley D. Gifford, Kamal Sharma, Martyn Goulding, Samuel L. Pfaff
宣传办公室
电话:(858) 453-4100
press@salk.edu
萨尔克研究所是一个独立的非营利性研究机构,由首个安全有效的脊髓灰质炎疫苗的研发者乔纳斯·索尔克于1960年创立。该研究所的使命是推动以合作、敢于冒险为特点的基础性研究,以应对癌症、阿尔茨海默病和农业脆弱性等社会最紧迫的挑战。这项基础科学支撑着所有的转化研究,产生有助于全球新药和创新的见解。.