
July 10, 2018
Salk-led study clarifies dual role of protein that watches for cellular threats and repairs damage
Salk-led study clarifies dual role of protein that watches for cellular threats and repairs damage
LA JOLLA—One reason we’re supposed to eat a variety of colorful fruits and vegetables is because they contain nutritious compounds called antioxidants. These molecules counteract the damage to our bodies from harmful products of normal cells called reactive oxygen species (ROS).
Now, research led by a Salk Institute professor along with collaborators from Yale, Appalachian State University and other institutions found that a protein called ATM (short for ataxia-telangiectasia mutated) can sense the presence of ROS and responds by sounding the alarm to trigger the production of antioxidants.
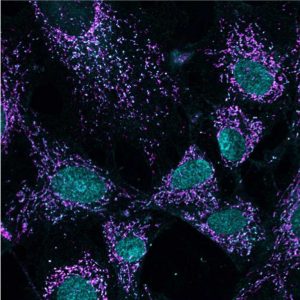
点击此处 用于高分辨率图像。.
版权:萨克研究所
The work, which appears in 科学信号学 on July 10, 2018, could have implications for a disease in which ATM is dysfunctional—and could also help reveal ways to boost cellular health overall.
“In ataxia-telangiectasia, the disease caused when the ATM is gene is mutated, people are prone to DNA damage because one of ATM’s functions is to repair DNA,” says Salk Professor 杰拉尔德·沙德尔, the paper’s co-corresponding author. “But we also see signs in this disease of damage caused by ROS, and it hasn’t been clear why that would be connected to dysfunctional ATM.”
Shadel studies mitochondria, the powerhouses of cells, which convert our food into chemical energy cells use. In the process, mitochondria produce the ROS that not only damage cells but also are danger signals. To better understand the role of ATM, Shadel began by investigating ATM’s response to ROS produced by mitochondria.
His team exposed laboratory cells in culture dishes to a chemical that encourages mitochondria to produce ROS. As expected, they saw increased ROS, but they also observed ATM molecules pairing up into what scientists call a dimer, which is not what ATM does when responding to DNA damage. These observations corroborate other research suggesting that ATM has two modes for responding to different types of cellular threats—DNA damage and ROS from mitochondria.
Treating the cells with a chemical that causes DNA damage did not induce ATM to form dimers, and the non-dimerized ATM went on to prompt damage-repair mechanisms. The scientists figured ATM’s formation of dimers in the presence of ROS represents a type of ROS-sensing function. Dimerized ATMs induced an entirely different mechanism than non-dimerized ATM: the pentose phosphate pathway (PPP), which is a series of biochemical steps that generates cellular antioxidants.

点击此处 用于高分辨率图像。.
版权:萨克研究所
ATM is like a smoke detector that also has a carbon monoxide sensor. Either a fire (DNA damage) or carbon monoxide (ROS) will cause the detector (ATM) to sound the alarm to protect your health.
“ATM is well known for its role in repair of DNA damage, but why it forms dimers in response to reactive oxygen species has been a mystery,” says co-corresponding author Brooke E. Christian of Appalachian State University. “This work is exciting because it reveals a functional consequence of ATM dimerization: to increase cellular antioxidant capacity through activation of the pentose phosphate pathway. It makes sense for ATM to have this function as a way to protect the genome from the damaging effects of reactive oxygen species.”
“We went into the study wanting to know the mechanism and function of the ATM-mediated mitochondrial ROS signaling pathway,” says Yichong Zhang, a graduate student researcher at Yale University and the paper’s first author. “The most exciting moment for me was when we discovered the details of the mechanism by which ROS signaling through ATM regulates cellular antioxidant responses.”
The revelation of how ATM and the production of antioxidants are connected via this pentose phosphate pathway could lead to ways to develop new treatments for the disease ataxia-telangiectasia.
Other authors included Ji-Hoon Lee and Tanya T. Paull of the University of Texas at Austin and the Howard Hughes Medical Institute; Sarah Gehrke and Angelo D’Alessandro of the University of Colorado, Denver; Qianhui Dou and Vadim N. Gladyshev of Harvard Medical School; Elizabeth A. Schroeder of Yale University; and Samantha K. Steyl of Appalachian State University.
The work was funded by NIH R01 AG047632 and R33 ES025636, the A-T Children’s Project, the Audrey Geisel Chair fund, the Joseph A. and Lucille K. Chair fund, NIH R01 GM065204, NIH NRSA NS077723, NIH F31AG043242, the China Scholarship Counsel and the Boettcher Foundation.
日记
科学信号学
作者
Yichong Zhang, Ji-Hoon Lee, Tanya T. Paull, Sarah Gehrke, Angelo D’Alessandro, Qianhui Dou, Vadim N. Gladyshev, Elizabeth A. Schroeder, Samantha K. Steyl, Brooke E. Christian and Gerald S. Shadel
期刊封面
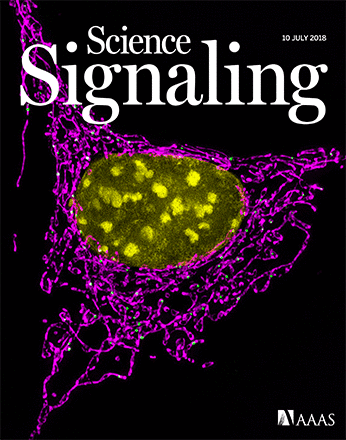
Online Cover This week features a Research Article that shows how the kinase ATM directs cell metabolism to prevent oxidative stress. The image shows fluorescent staining of mitochondria and the nucleus in NIH 3T3 cells. [Image: Uri Manor, Salk Institute for Biological Studies]
宣传办公室
电话:(858) 453-4100
press@salk.edu
萨尔克研究所是一个独立的非营利性研究机构,由首个安全有效的脊髓灰质炎疫苗的研发者乔纳斯·索尔克于1960年创立。该研究所的使命是推动以合作、敢于冒险为特点的基础性研究,以应对癌症、阿尔茨海默病和农业脆弱性等社会最紧迫的挑战。这项基础科学支撑着所有的转化研究,产生有助于全球新药和创新的见解。.