
August 14, 2014
Salk scientists find control signal for immune system that could help treat autoimmune diseases and cancer.
Salk scientists find control signal for immune system that could help treat autoimmune diseases and cancer.
LA JOLLA–When faced with pathogens, the immune system summons a swarm of cells made up of soldiers and peacekeepers. The peacekeeping cells tell the soldier cells to halt fighting when invaders are cleared. Without this cease-fire signal, the soldiers, known as killer T cells, continue their frenzied attack and turn on the body, causing inflammation and autoimmune disorders such as allergies, asthma, rheumatoid arthritis, multiple sclerosis and type 1 糖尿病.
Now, scientists at the Salk Institute have discovered a key control mechanism on the peacekeeping cells that determine if they send a halt signal to the killer T cells. The new research, published today in 细胞, could help develop treatments for autoimmune disorders as well as some types of 癌症.
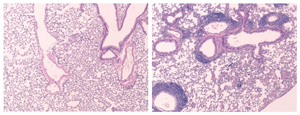
In lungs lacking a ‘halt’ signal from regulatory T cells, (right) killer T cells and other pathogen fighters accumulate (purple rings) in an excessive immune system response, creating lung inflammation and asthma-like symptoms.
点击此处 用于高分辨率图像。.
图片:由萨克生物研究所提供
“We discovered a mechanism responsible for stabilizing the cells that maintain immune system balance,” says senior author 叶正, Salk assistant professor and holder of the Hearst Foundation Developmental Chair. “The immune system plays a huge role in chronic inflammation and if we can better understand the immune system, we can start to understand and treat many diseases.”
This balance of signaling for the peacekeeping white blood cells–known as regulatory T cells (“Tregs”)–is crucial to normal immune response. Aside from letting killer T cells run rampant, Tregs can conversely send too many cease-fire signals, which causes killer T cells to ignore threatening invasions. For example, some tumors nefariously surround themselves with a high density of Tregs transmitting the cease-fire signal to protect themselves from being attacked.
“Tregs are like the surveillance system of the immune response,” says Zheng. “This surveillance system is key to healthy immune reactions, but it can be kicked into overdrive or turned entirely off.”
For about a decade, researchers knew that the key to Tregs’ peacekeeping ability is a gene called Foxp3, but they weren’t sure how exactly it worked. Researchers also knew that under certain conditions, Tregs can go rogue: they transform into killer T cells and join in the siege. This change means that they lose the ability to send a ‘halt’ signal and add to inflammation.

(From left) Xudong Li, Ye Zheng and Yuqiong Liang of Salk’s Nomis Foundation Laboratories for Immunobiology and Microbial Pathogenesis
点击此处 用于高分辨率图像。.
图片:由萨克生物研究所提供
In the new paper, Zheng’s lab reports that a particular genetic sequence in Foxp3 is solely responsible for the stability of a Treg. If they removed the sequence, dubbed CNS2, Tregs became unstable and often morphed into killer T cells–the type of cell they are supposed to be controlling–resulting in autoimmune disease in animal models.
“Foxp3 safeguards Treg to not become anything else,” says Zheng. “Previously, very little was known on how Foxp3 did this. We discovered the area of the Foxp3 gene that determines the stability of Tregs and keeps the immune system balanced.”
Without this specific region in Foxp3, Treg cells are much more likely to lose their identity and defect into killer T cells when faced with inflammation and infection, says Zheng.
He adds that recent new drugs on the market or in clinical trials are attempting to disable Tregs in tumors to help the body’s own immune system fight cancer. This new work provides a target for future cancer drugs as well as autoimmune treatments.
“Now we can try to target this region on Foxp3 to either enhance or reduce the impact of Tregs for treatment of autoimmune disease or cancer, respectively,” Zheng adds.
Authors of the work also include Salk researchers Xudong Li, Yuqiong Liang, Mathias LeBlanc and Chris Benner. Funding was provided by the 美国国立卫生研究院, , 那个 Nomis Foundation, , 那个 National Multiple Sclerosis Society, , 那个 丽塔·艾伦基金会, the Emerald Foundation, the Hearst Foundation, and the James B. Pendleton Charitable Trust.
关于索尔克生物研究所:
索尔克生物学研究所是世界上首屈一指的基础研究机构之一,其国际知名的教职员工在一个独特的、协作和创新的环境中,深入探索生命科学的基本问题。索尔克科学家既注重科学发现,也注重培养下一代研究人员,通过研究神经科学、遗传学、细胞生物学、植物生物学及相关学科,为我们理解癌症、衰老、阿尔茨海默病、糖尿病和传染病做出了开创性的贡献。.
教职员工的成就获得了无数荣誉,包括诺贝尔奖和美国国家科学院院士资格。该研究所由脊髓灰质炎疫苗先驱 Jonas Salk 医生于 1960 年创立,是一家独立的非营利组织和建筑地标。.
日记
细胞
作者
Xudong Li, Yuqiong Liang, Mathias LeBlanc, Chris Benner and Ye Zheng
宣传办公室
电话:(858) 453-4100
press@salk.edu