
August 25, 2016
Salk researchers have succeeded in, for the first time, creating kidney progenitor cells that survive in the lab
Salk researchers have succeeded in, for the first time, creating kidney progenitor cells that survive in the lab
点击此处 用于高分辨率图像
版权:萨克研究所
LA JOLLA—Salk Institute scientists have discovered the holy grail of endless youthfulness—at least when it comes to one type of human kidney precursor cell. Previous attempts to maintain cultures of the so-called nephron progenitor cells often failed, as the cells died or gradually lost their developmental potential rather than staying in a more medically useful precursor state.
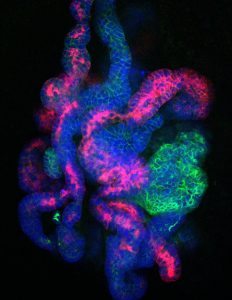
But by using a three-dimensional culture and a new mixture of supporting molecules, Salk researchers have successfully suspended the cells early in their development. Such early-stage kidney cells could be used to grow replacement kidney tissue in order to study the organ as well as treat disease.
“We provide a proof-of-principle for how to make and maintain unlimited numbers of precursor kidney cells,” says 胡安·卡洛斯·伊兹皮苏亚·贝尔蒙特, professor in Salk’s Gene Expression Laboratory. “Having a supply of these cells could be a starting point to grow functional organs in the laboratory as well as a way to begin applying cell therapy to kidneys with malfunctioning genes.” The work was published in Cell Stem Cell on August 25, 2016.
Nephron progenitor cells (NPCs), at least in humans, normally only exist during a brief stage of embryonic development. The cells go on to form nephrons, the functional units of the kidney, responsible for filtering the blood and excreting urine. But adults have no remaining NPCs to grow new kidney tissue after damage or disease. Generating NPCs in the lab, scientists believe, will offer a new way to study kidney development and eventually treat kidney diseases.
点击此处 用于高分辨率图像
版权:萨克研究所
Previously, other groups of scientists have used induced pluripotent stem cells (iPSCs) to make NPC-like cells. “Those approaches take a long time, it is difficult to isolate a pure population, and the NPC-like cells are still transient,” says Zhongwei Li, a research associate in the Izpisua Belmonte lab and co-first author of the new paper. In those cases, the NPCs often matured into adult kidney cells in a manner of days, leaving no steady population of progenitor cells to study.
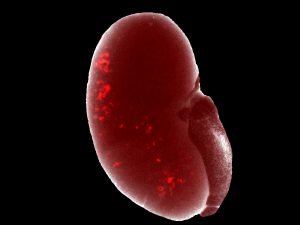
First working with NPCs directly isolated from mouse embryos, Izpisua Belmonte, Li and collaborators worked to develop methods that would keep the NPCs in their usually transient, progenitor state. They discovered that if they maintained the cells in a three-dimensional culture, rather than a flat dish, and used a new mixture of signaling molecules, they could maintain the NPCs for more than 15 months. They went on to show that the cells—when moved to new conditions—could then be coaxed to develop into functional nephron-like structures both in the lab or when transplanted into animals.
Next, the team used both human embryonic NPCs and human NPCs generated from stem cells to tweak the protocol for human use. Again, they were able to maintain the NPCs long term.
“The 3D culture strategy used in our study can potentially be applied to other lineage progenitors for efficient formation of tissue organoids,” says co-first author Jun Wu, Salk research associate.

点击此处 用于高分辨率图像
版权:萨克研究所
Aside from a regenerative therapy to replace ailing organs, the scientists add that the NPCs could be used to model diseases in the lab. By introducing disease related mutations to the cells, researchers could study the onset and progression of the disease and gain new insights into the disease as well as screen and discover new treatment drugs.
Next, the researchers would like to investigate how to culture the other types of progenitor cells that are required for a full kidney, in addition to the nephrons formed by NPCs. “There are several progenitor cells that work together to make a whole organ,” adds co-first author Toshikazu Araoka, Salk research associate. “If we can culture the other progenitor cells as well, we’ll be closer to building a transplantable kidney.”
Other researchers on the study were Hsin-Kai Liao, Mo Li, Min-Zu Wu, Isao Tamura, Yun Xia, Ergin Beyret, and Concepcion Rodriguez Esteban of the Salk Institute; Marta Lazo and Josep M. Campistol of the Hospital Clinic of Barcelona; Bing Zhou and Yinghui Sui of the 加州大学圣地亚哥分校; Taji Matsusaka of Tokai University School of Medicine; Ira Pastan of the 国家癌症研究所; Isabel Guillen and Pedro Guillen of Clinica Cemtro in Madrid.
The work and the researchers involved were supported by grants from the Kyoto University Foundation, the Nagai Foundation Tokyo, the Kidney Foundation Japan, the 加州再生医学研究所, a Catharina Foundation fellowship, the Universidad Católica San Antonio de Murcia (UCAM), Fundacion Dr. Pedro Guillen, G.哈罗德和莱拉·Y·马瑟斯慈善基金会, 利昂娜·M·哈里·B·赫尔姆斯利慈善信托, ,和 The Moxie Foundation.
日记
Cell Stem Cell
作者
Zhongwei Li, Toshikazu Araoka, Jun Wu, Hsin-Kai Liao, Mo Li, Marta Lazo, Bing Zhou, Yinghui Sui, Min-Zu Wu, Isao Tamura, Yun Xia, Ergin Beyret, Taiji Matsusaka, Ira Pastan, Concepcion Rodriguez Esteban, Isabel Guillen, Pedro Guillen, Josep M. Campistol, Juan Carlos Izpisua Belmonte
宣传办公室
电话:(858) 453-4100
press@salk.edu
萨尔克研究所是一个独立的非营利性研究机构,由首个安全有效的脊髓灰质炎疫苗的研发者乔纳斯·索尔克于1960年创立。该研究所的使命是推动以合作、敢于冒险为特点的基础性研究,以应对癌症、阿尔茨海默病和农业脆弱性等社会最紧迫的挑战。这项基础科学支撑着所有的转化研究,产生有助于全球新药和创新的见解。.